 Reversibility requires the direction of heat transfer to reverse for the reverse path. So, for example, if the path BA is followed and there is friction, then the gas will be returned to its original state but the environment will not-it will have been heated in both directions. If there are any energy-dissipating mechanisms, such as friction or turbulence, then heat transfer to the environment occurs for either direction of the piston. In the previous examples, our system is a gas (like that in ), and its environment is the piston, cylinder, and the rest of the universe. Real macroscopic processes are never exactly reversible. The reverse isothermal and adiabatic paths are BA and CA, respectively. A reversible process is one in which both the system and its environment can return to exactly the states they were in by following the reverse path. If the path ABCA could be followed by cooling the gas from B to C at constant volume (isochorically), (b), there would be a net work output.īoth isothermal and adiabatic processes such as shown in are reversible in principle. Lower temperature results in lower pressure along the way, so that curve AC is lower than curve AB, and less work is done. Since the pressure is constant, the force exerted is constant and the work done is given as 
PV Diagrams and their Relationship to Work Done on or by a GasĪ process by which a gas does work on a piston at constant pressure is called an isobaric process. In this section, we consider some of the simpler underlying processes on which heat engines are based. We will examine heat engines in detail in the next section. Variations of this process are employed daily in hundreds of millions of heat engines. Heat transfer now occurs from the gas to the surroundings so that its pressure decreases, and a force is exerted by the surroundings to push the piston back through some distance. To repeat this process, the piston needs to be returned to its starting point. Heat transfer to the gas cylinder results in work being done. The gas does work on the outside world, as this force moves the piston through some distance. Fuel combustion produces heat transfer to a gas in a cylinder, increasing the pressure of the gas and thereby the force it exerts on a movable piston. 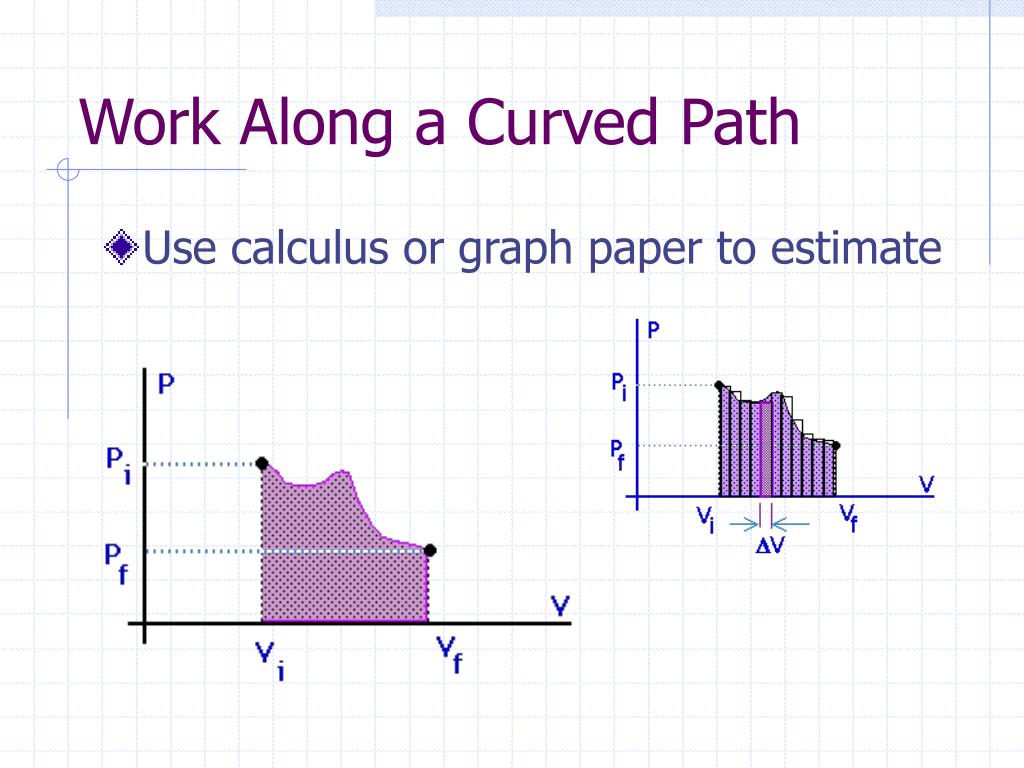
The illustrations above show one of the ways in which heat transfer does work. shows schematically how the first law of thermodynamics applies to the typical heat engine. Car engines and steam turbines that generate electricity are examples of heat engines. 
One of the most important things we can do with heat transfer is to use it to do work for us.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
